Back in December 2015, parties to the United Nations Framework Convention on Climate Change reached a landmark agreement. One hundred and ninety-five nations, practically every country in the world, were going to officially fight against the climate crisis. This is now known as the Paris Agreement. And the main goal of that treaty is to limit global temperature rise to well below two degrees Celsius.
But in order to save the Earth from rising temperatures, we need to remove billions of metric tons of carbon from our atmosphere every year. To help accomplish this, countries have been investing in negative emissions and low-carbon technologies, as well as adding infrastructure to support them. And solar energy is already at the forefront of this renewable revolution. Why? Because the sun is the most abundant energy resource we have on our planet. But how much do you know about solar panels and how exactly do they work?
Well, let's start with photovoltaic cells.  These cells are what convert sunlight into electricity. A single photovoltaic cell, also called a solar cell, can be smaller than a postage stamp and thinner than a human hair.
These cells are what convert sunlight into electricity. A single photovoltaic cell, also called a solar cell, can be smaller than a postage stamp and thinner than a human hair.
On its own, a solar cell can generate about half a volt. So, to increase this energy output, you can combine these solar cells to create solar modules and even slightly-bigger solar panels.
Depending on their size and the materials they're made of, as well as the amount of sunlight that's available, the power output of solar panels can vary. This output, or the amount of energy a panel produces, is measured in kilowatt-hours. So, if a panel generates 100 watts in one hour, that would be 100 watt-hours or 0.1 kilowatt-hours.
But you can combine modules and panels even further to create solar arrays. These structures are what are used to power homes and even spacecraft. And if you gather enough solar arrays in one place, you can even power cities.
You may have seen swaths of land covered in solar arrays. These are often called solar farms or solar parks. The largest solar park built to date is the Pavagada Solar Park in India. It spans over 53 square kilometers and can produce two gigawatts of electricity, which is enough to power 700,000 households. By the end of 2018, the world's installed capacity of solar cells reached over 480 gigawatts, representing the second largest renewable electricity source after wind.
By the end of 2018, the world's installed capacity of solar cells reached over 480 gigawatts, representing the second largest renewable electricity source after wind.
Now, this may seem like a lot of energy already, but researchers are projecting that by 2050 the world's solar cell capacity could reach over 8,500 gigawatts. This is the kind of expansion we need to reach the carbon-cutting goals of the Paris Agreement.
The big reason why photovoltaic cells haven't taken over the world yet is because the technology is still limited by three important factors. Cost, efficiency and reliability.
Efficiency basically just means how well a solar panel is able to convert sunlight into electricity, and cost can be defined by, well, how much goes into making a solar cell like materials, manufacturing, distribution, installation, relative to how much wattage is generated in return for that investment. And lastly, we have to make sure a solar cell can generate power on sunny and cloudy days, which is tougher than you might think.
In recent years, advances to solar energy technology have helped it become more accessible to the average person like you and me, and to the teams taking part in the World Solar Challenge.
So, basically, the sun provides energy in the form of photons. And when those photons will hit your solar ray when it hits your solar cells, the photons excite electrons and those will jump across the band gap inside your solar cell and they'll basically flow through your circuit. So, you get this flow of electrons.
Today, the most popular semiconducting material used in solar cells is silicon. Diving in even deeper, we'll see that crystalline silicon cells are made of silicon atoms connected to one another to form a crystal lattice.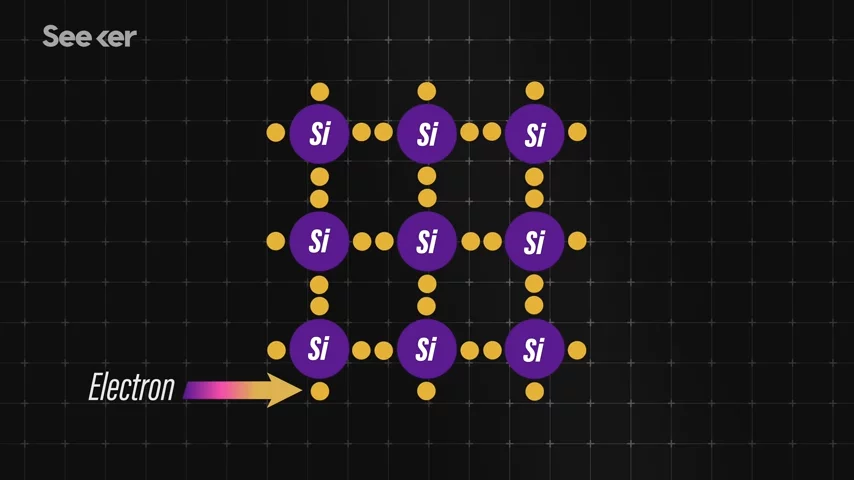 Silicon bonds are made of electrons, the negatively-charged particles in an atom. These electrons allow it to perfectly bond to its silicon neighbors, creating this perfectly organized lattice structure.
Silicon bonds are made of electrons, the negatively-charged particles in an atom. These electrons allow it to perfectly bond to its silicon neighbors, creating this perfectly organized lattice structure.
In a solar cell, there are two layers of silicon. One layer, n-type, has a negative charge, and the other layer, p-type, has a positive charge.  Now, each charge needs to be enhanced to create the energy we're looking for. And to do that, researchers will dope or add other elements to the silicon material, giving it extra electrons, or creating empty holes for electrons to fill.
Now, each charge needs to be enhanced to create the energy we're looking for. And to do that, researchers will dope or add other elements to the silicon material, giving it extra electrons, or creating empty holes for electrons to fill.
The negative charge is usually achieved by mixing the layer of silicon with phosphorus. This adds extra electrons to the mix, allowing more electrons to roam freely in the lattice. 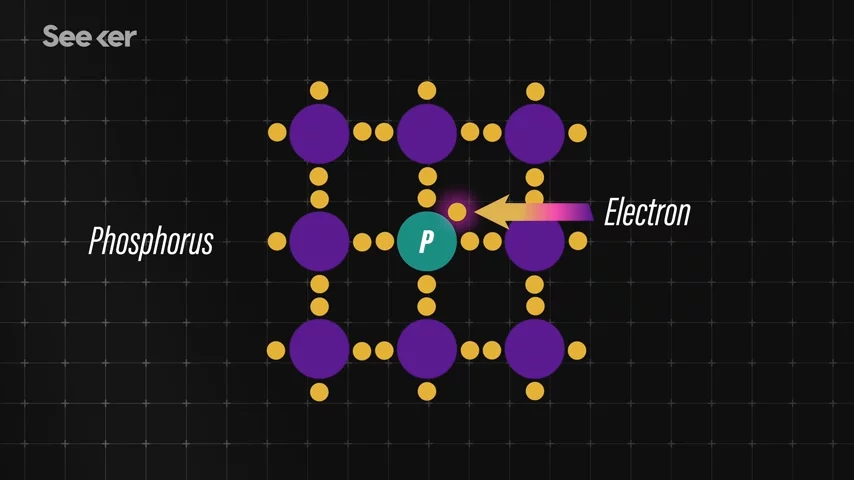 A positive charge for that p-layer is achieved by doping that layer with boron, causing those spaces called holes.
A positive charge for that p-layer is achieved by doping that layer with boron, causing those spaces called holes.
The boundary between the two layers is called the p-n junction, while the area around it is known as the depletion region. So, now for the fun part. When light from the sun hits those layers, the energy from the photons knocks electrons loose.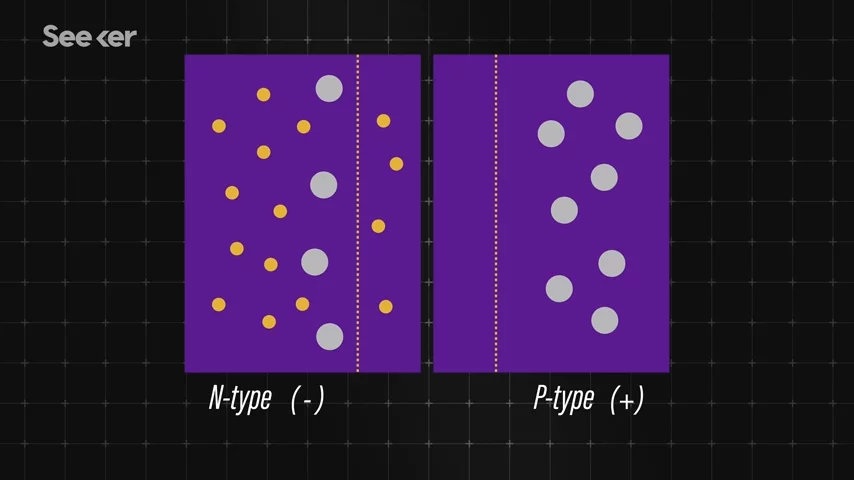 Because the layers are oppositely charged, the electrons want to travel from the n-type layer to the p-type layer to fill its empty holes.
Because the layers are oppositely charged, the electrons want to travel from the n-type layer to the p-type layer to fill its empty holes.
The electrons create a voltage difference between either end of the cell. So, by adding an electric circuit to one end, the electrons can travel through that circuit, powering devices along their way and end up in the p-type layer.
But typical crystalline silicon PV cells only convert 18 to 22% of sunlight into electricity. And that's clearly not enough.
We want solar cells to be as efficient as possible, so they can power as many things as possible. And the teams competing in the World Solar Challenge are already using an advanced material to do so.
The typical material to make solar cells out of is silicon. They're allowed up to four square meters. But recently a lot of cars air switching over to a different material, gallium-arsenide cells, and they're significantly more efficient.
Gallium-arsenide is a semi-conducting material made from the elements gallium and arsenic. Gallium-arsenide solar cells now hold the world efficiency record for a single junction solar cell, with a conversion rate of just around 28.8%. So what's stopping us from using that in everything? Well, making a wafer of gallium-arsenide is considerably more expensive than making a silicon wafer.
The size of the gallium-arsenide array was 3.56 square meters, and to cover that size array, you need about 100 grand, um, to cover the same amount of area, actually to cover about four square meters. So even a larger area of silicon, you'll need about 3 grand, so there's a huge price difference.
So researchers are still trying to find that perfect solar cell that is both cost-effective and full efficiency. Innovations like those used in the World Solar Challenge are going to continue to push the boundaries of renewable solar technology. And now that you have a solid grasp on how solar panels and photovoltaics work, let's take a closer look at how we can apply all of this solar tech to a race car.


No comments yet