You know that batteries are complicated. They come in all shapes, sizes, charge capabilities, and we use them in everything. And if there's one battery technology that sets the gold standard, it's the lithium-ion battery.
These batteries are one of the only types to pack powerful energy storage in a small and lightweight design. So, when designing battery technologies for the future, the challenge is to improve upon the advantages that the lithium-ion battery already has. Because even though lithium-ion batteries currently dominate the global battery market, there are quite a few things about them that could be improved, like power output, energy capacity, cost, lifespan, and safety.
Now some of the most promising designs for giving lithium-ion battery technology a boost are the lithium-silicon battery, the lithium-sulfur battery, solid-state lithium-ion batteries and one that's a common nation of approaches. And changing out the materials used to make anodes, cathodes and electrolytes is exactly what scientists have been doing. But what is it going to take for one of these to become the new battery of the future?
It's important to note that a simple change in a battery's design can significantly affect its voltage and storage capacity. Here I've made a really simple homemade battery with 14 cells using an ice cube tray, steel screws, copper wire, a couple electrical leads, and this voltmeter. And the way this works is that each little ice cube tray is its own battery cell. So you've got the copper acting like the cathode. It's gaining electrons. The steel is acting as the anode. It's losing electrons. And the salt water is acting as my electrolyte, allowing that flow of charges.
Here I've made a really simple homemade battery with 14 cells using an ice cube tray, steel screws, copper wire, a couple electrical leads, and this voltmeter. And the way this works is that each little ice cube tray is its own battery cell. So you've got the copper acting like the cathode. It's gaining electrons. The steel is acting as the anode. It's losing electrons. And the salt water is acting as my electrolyte, allowing that flow of charges.
When I hook it up to the voltmeter, you can see I am reading a voltage of around 200 millivolts. Now, what I'm interested in is what would happen if we changed out the electrolyte solution to the, say, vinegar.  Lemon juice.
Lemon juice.  So all these sorts of things can make a significant impact on the voltage and the storage capacity of the battery.
So all these sorts of things can make a significant impact on the voltage and the storage capacity of the battery.
First up, silicon anode batteries. Remember that anodes are the negative electrode within a call? Well, like we talked about in the previous article, the current most popular anode material in lithium-ion cells is graphite. This is because graphite's structure helps keep those lithium ions efficiently stored in the anode. But there is a maximum amount of lithium-ions that can be stored in the anode, and that determines the cell's capacity. And as it turns out, silicon does a much better job than graphite at absorbing and holding lithium-ions.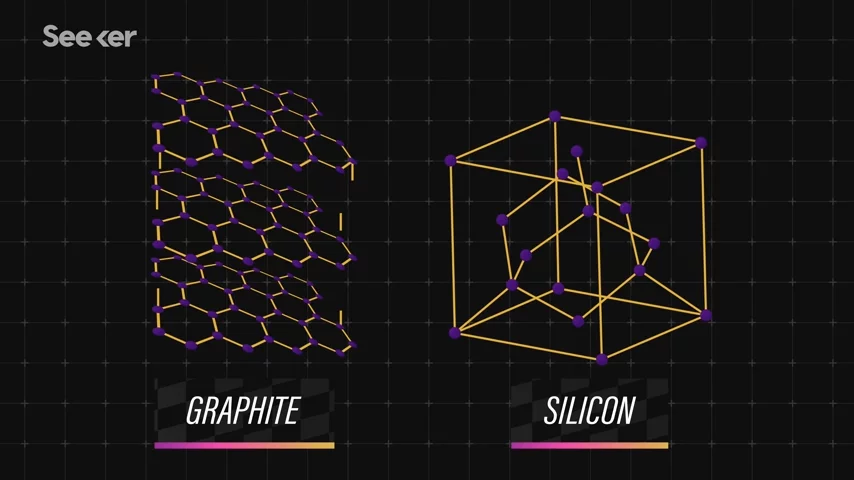 And this means batteries can be made smaller, more energy-efficient, and cheaper. But, of course, this does all come with a catch.
And this means batteries can be made smaller, more energy-efficient, and cheaper. But, of course, this does all come with a catch.
Silicon anodes have a tendency to dramatically expand when encountering lithium during charging. And those anodes also then shrink when the battery discharges. And this repeated expansion and contraction shortens the lifespan of the battery, and ultimately, its usefulness. But researchers like those at Enovix, are aiming to fix this problem.
We don't eliminate anode expansion and contraction, but we do control them. Propendent 3D cell architecture enables us to integrate a very thin stainless steel constraint into our battery design.
This applies a uniform force around the battery to constrain the silicon expansion within the cell. during the charging cycles and during discharge.
While some researchers have set their sights on the anode, others are experimenting with the cathode with one innovation being lithium sulfur cells.
Lithium on its own is a very volatile substance. It reacts to air, it reacts to water. So what the OXIS scientists have done is taken sulfur as a non-conductive, very cheap material. and used the sulfur to act as a fire retardant around lithium metal, so that if air or water impacts lithium metal, thermal runaway, fire, explosion, doesn't take place.
Sulfur cathodes, like their silicon anode counterparts, can absorb more lithium ions than the typical cobalt-based cathodes. Offering a reduced battery cost with increased energy density and improved safety compared to lithium-ion batteries.
Because one of the key factors of lithium sulfur is that it is 50 to 60% lighter than lithium-ion. If you take a bus with a very large battery, if you can replace that technology with lithium-sulfur, and you reduce the weight and still extend the distance covered, then you have a major breakthrough in the renewable transportation systems.
But lithium-sulfur cells are still not quite perfect because they face the challenge of lithium-polysulfide formation, or what's known as the polysulfide shuttle. The sulfur electrode also expands and contracts as it cycles, which results in a loss of battery efficiency and power and energy density. But what if the answer isn't in the anode or the cathode? What about the electrolyte?
Well, that's where solid state batteries come in. Solid state electrolytes have been around for a while and have recently caught on as a contender for future batteries because of their promise of improved safety. But solid state polymers can better withstand extreme conditions. So when heated, they behave like liquids, but they can operate without the danger of bursting into flames.
Some researchers believe that solid state batteries could even give electric vehicles over 500 miles of range. And if we really let our imaginations run wild, using solid state batteries in solar-powered vehicles like the ones that compete in the World Solar Challenge could potentially lead to even longer ranges.
Now, what's the downside to these solid state batteries? Well, unlike liquid electrolytes, they can't stay in contact with every bit of the electrodes all the time. And this makes it harder for the ions to move between electrodes and create that flow of electricity that we need. But what if we were able to combine a few of these innovations that we've already talked about?
We could now make a transition from liquid electrolyte to solid state lithium sulfur. And I'm talking about removing the diesel trucks, diesel buses, the lead based fuel that our aircraft consume. These are the biggest pollutants that we've got on the planet, and solid state is certainly the phenomenon that will render those achievements more realistic.
So OXIS Energy is currently not in the solar car market, but is instead focusing on aerospace, marine vessels, and electric vehicles. They're close to achieving an energy density of 500 watt-hours per kilogram with their battery, and have already set a new target of 600 watt-hours per kilogram.
Essentially, that means a battery like this in the future could be capable of powering an electric car for 1,000 kilometers on a single charge. By comparison, Tesla's Panasonic lithium-ion battery cells, which are currently the most commercially advanced, are about half as energy dense. So all of the battery innovations we've covered are definitely impressive. But if we want more solar vehicles on the roads, we're going to need a powerful battery storage system with high energy density, high efficiency, and the ability to last long on the road, rain or shine, because currently, none of the options on the market or even in development totally do the job.


No comments yet